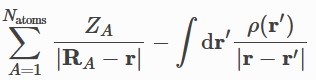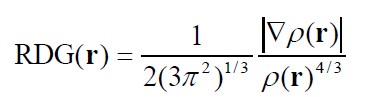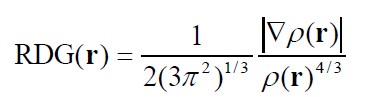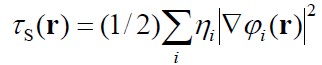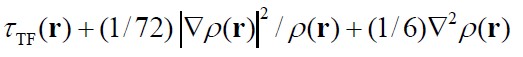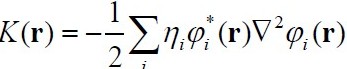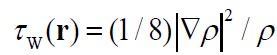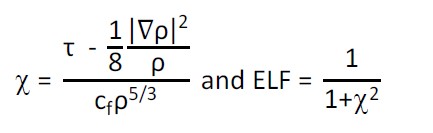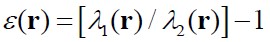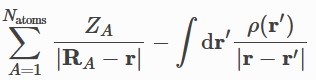Current local descriptors computed at r =
(x, y, z)
- Electron density ρ(r).
- Components of electric dipole moment, - x ρ(r); - y ρ(r); - z ρ(r)
- Radial distribution, 4 π
r2 ρ(r) with (r2 = x2 + y2+ z2).
- Gradient of electron
density ∇ρ(r) its norm |∇ρ(r) |.
- Eigenvalues of the Hessian matrix of electron density : λ1(r) , λ2(r) and λ3(r).
- Laplacian of electron density ∇2ρ(r) = λ1 + λ2 + λ3 ; L(r) = - ∇2ρ(r).
- Shape function, ρ(r)/N,
where N is the total number of electrons.
- Reduced Density Gradient (RDG), see J. Am. Chem. Soc., 132, 6498 (2010).
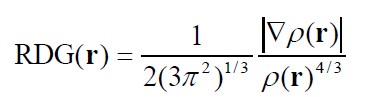
- τs(r) or τ(r) or G(r) is the exact kinetic energy
density (KED) of the
non-interacting electron system defined within the KS-DFT theory.
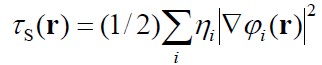
where φi
are the occupied molecular orbitals.
- Local Temperature (2/3) G(r)/ρ(r) in Hartree/kB. see PNAS, 81, 8028 (1984)
- KED per electron G(r)/ρ(r).
- Approximate Kirzhnits G(r),
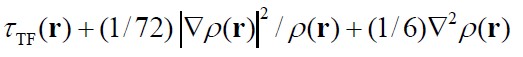
with τTF(r)
is the Thomas-Fermi KED, exact KED of an uniform electron gas
(non-interacting system). The degree of ELF localization is given with
respect to theThomas-Fermi localized degree.
- Hamiltonian K(r) kinetic
energy density
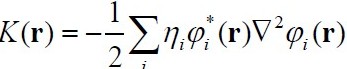
- Potential energy density
(Virial field): V(r) = - [ G(r) + K(r) ] = 0.25 ∇2ρ(r) - 2 G(r)
- Ratio V(r)|/G(r).
At the QTAIM bond critical bond, ratio<1 for a closed-shell
interaction and a ratio >1 for a typical covalent bond. See J. Chem. Phys., 117, 5529 (2002).
- Ratio |V(r)|/(2G(r)). At the QTAIM bond critical
bond, ratio> for a typical covalent bond.
- Total energy H(r) = G(r) + V(r) = - K(r)
- Weizsäker kinetic energy density τw(r)
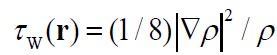
τw(r) is
the exact kinetic energy density of any one-orbital system interpreted
as a steric energy density. See J.
Chem. Phys., 126, 244103 (2007)
- Electron Localization Function (ELF)
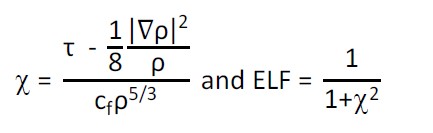
ELF definition for
HF/Kohn-Sham DFT. See Angew. Chem.
Int. Ed. Engl., 31, 187 (1992). ELF reveals the atomic shell
structure as well as the nature of intra- and intermolecular
interactions.
- Approximate ELF computed from the Kirzhnits approximation
of G(r). See Chem. Phys. Lett., 351, 142 (2002).
- Ellipticity,
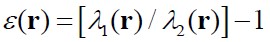
where λ1 and λ2
are the lowest and the second
lowest eigenvalues of the electron density, respectively. This
descriptor reveals an asymmetric distribution of the electron density
around the QTAIM bond critical bond.
see J. Am. Chem. Soc., 105, 5061 (1983).
- Bond metallicity,
ρ(r)/∇2ρ(r). Indicates the metallic
interaction computed at the BCP. see J.
Phys.: Condens. Matter, 14, 10251 (2002).
- Shannon Entropy Density, - ρ(r)
ln(ρ(r)). See J. Chem. Phys., 126, 191107 (2007).
- Molecular Electrostatic
Potential MESP, electron & nuclear parts of the MESP.